Falsified Mounjaro pens prompt urgent safety advisory
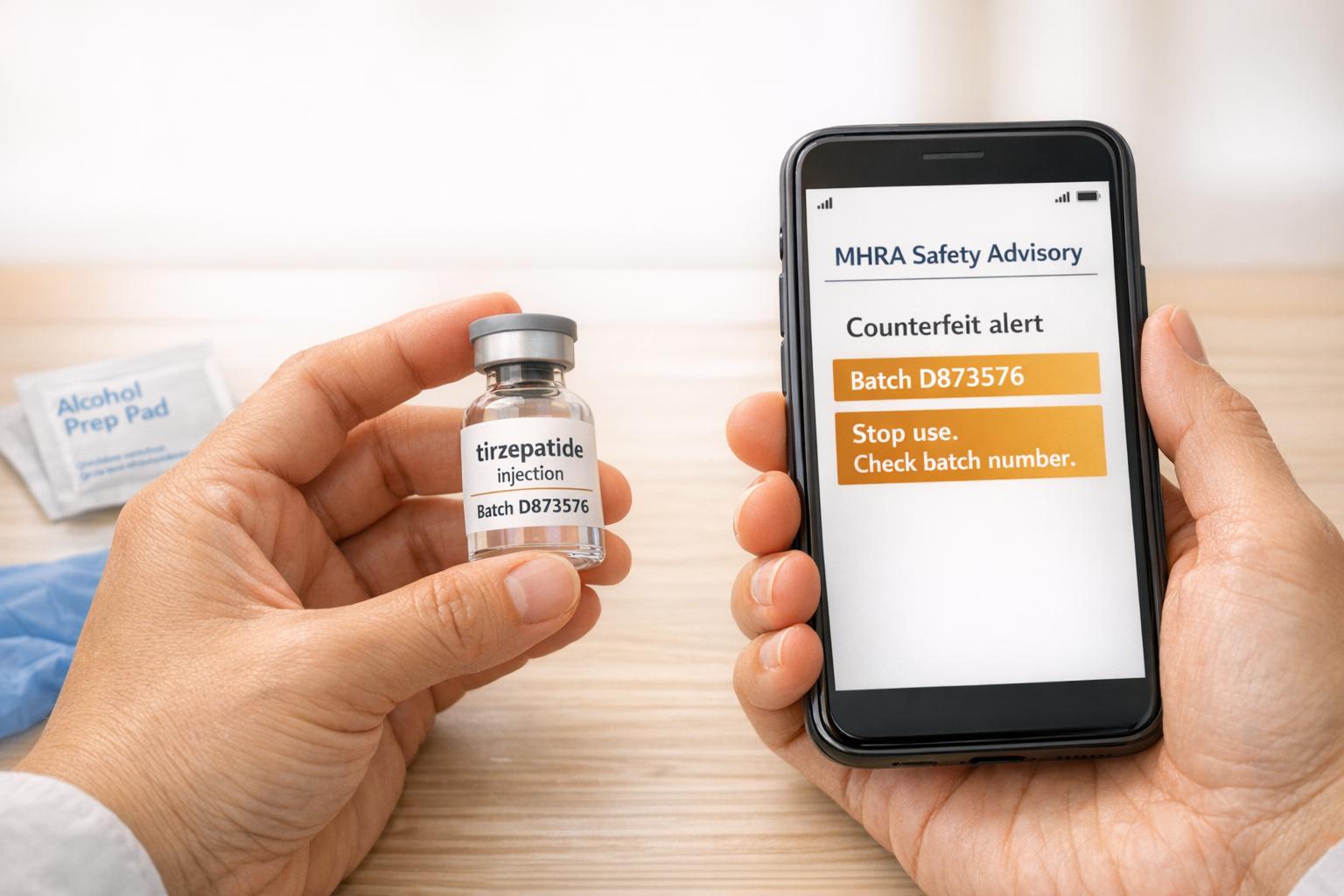
A warning has been issued following the discovery of counterfeit Mounjaro pens distributed by an online pharmacy. The Medicines and Healthcare products Regulatory Agency (MHRA) has urged users to check for specific batch numbers to ensure their safety.
The counterfeit product in question is the Mounjaro (tirzepatide) KwikPen 15mg, identified by the batch code D873576. According to the MHRA, the pens were supplied by the Private Pharmacy Clinic in Birmingham, with five of the affected pens confirmed to have been distributed. While testing showed that the pens do contain tirzepatide – the active substance in Mounjaro – their manufacturing conditions remain unknown, raising concerns about sterility.
As the product is injectable, the lack of confirmed sterility presents potential risks, including infections and allergic reactions. Symptoms of infection may include redness, swelling, or warmth at the injection site, pain, fluid leakage, a mild fever, or chills, among others. Allergic reactions could manifest as a rash, itching, or breathing difficulties. Anyone experiencing these symptoms should seek medical attention immediately.
The issue was brought to the attention of the MHRA by Eli Lilly and Company Limited, the legitimate manufacturer of Mounjaro. Defects with the pens were reported, with the dose knob detaching during use in all but one of five documented cases. Dr. Alison Cave, Chief Safety Officer at the MHRA, noted: "Check the batch number and strength of your KwikPen. If you have one of the affected pens, stop using it straight away. Please report it to the MHRA by emailing [email protected]. Please include ‘Mounjaro Pens’ in the subject line. Keep the pen in a safe place."
Dr. Cave reassured users that the immediate risk is considered low, stating: "If you have administered injections using the pen already, please be reassured that, based on the cases reviewed to date, the risk to you is low. Testing has confirmed that the identified fake pens do contain tirzepatide, the active substance in Mounjaro, but because the manufacturing conditions are unknown, we cannot confirm that the contents are sterile."
The MHRA advises individuals unsure about identifying their pens or determining if they’ve used an affected product to consult a healthcare professional. Suspected counterfeit medications or defective pens should also be reported through the MHRA’s Yellow Card scheme.
Mounjaro is a prescription injectable medication approved for weight management. However, vigilance is critical when securing such products. For users in possession of Mounjaro pens, verifying batch numbers and seeking guidance if necessary is strongly encouraged.
Read the source

Transforming Lives, One Step at a Time
Keep reading
How to Adjust Weight Loss Goals on GLP-1 Medications
Adjust weight goals on GLP-1 meds by tracking body composition and labs, optimizing protein, activity, sleep, and working with your provider on dosing.
Novo Nordisk shares drop as Alzheimer’s hopes for weight-loss drug fade
Novo Nordisk’s semaglutide failed to slow Alzheimer’s in large trials, prompting shares to fall and expert reactions.
China’s Supreme Court Upholds Semaglutide Patent for Novo Nordisk
China’s Supreme People’s Court upholds Novo Nordisk’s semaglutide compound patent, supporting IP protection.



